Visual or manual artifact rejection
Introduction
This tutorial makes use of the preprocessed data from the preprocessing tutorial. Run the script from that section in order to produce the single trial data structure, or download PreprocData.mat. Load the data into MATLAB memory with the following command:
load PreprocData data_all
Background
For a successful analysis of EEG or MEG signals you want to have “clean” data. That means that you should try to reduce the variance in the data due to factors that you cannot influence. One of those factors is the presence of artifacts in the data. These artifact can be physiological, can relate to the behaviour of the subject, or can result from the acquisition electronics or environmental sources. The strongest physiological artifacts stem from eye blinks, eye movements and head movements. Muscle artifact from swallowing and neck contraction can be a problem as well. Artifacts related to the electronics are for example ‘SQUID jumps’ in MEG.
In general it is best to avoid artifacts during the recording. You can instruct the subject not to blink during the trial, and instead give him some well-defined time between the trials in which he is allowed to blink. But of course there will always be some artifacts in the raw data.
While detecting artifacts by visual inspection, keep in mind that it is a subjective decision to reject certain trials and keep other trials. Which type of artifacts should be rejected depends on the analysis you would like to do on the clean data. If you would like to do a time-frequency analysis of power in the gamma band it is important to reject all trials with muscle artifacts, but for a ERF analysis it is more important to reject trials with drifts and eye artifacts.
In visual artifact detection, the user visually inspects the data and identifies the trials or data segments and the channels that are affected and that should be removed. The visual inspection results in a list of noisy data segments and channels.
The functions that are available for visual artifact detection are
The ft_rejectvisual function works only for segmented data (i.e. trials) that have already been read into memory. It allows you to browse through the large amounts of data in a MATLAB figure by either showing all channels at once (per trial) or showing all trials at once (per channel) or by showing a summary of all channels and trials. Using the mouse, you can select trials and/or channels that should be removed from the data. This function directly returns the data with the noise parts removed and you don’t have to call ft_rejectartifact or ft_rejectcomponent.
The ft_databrowser function works both for continuous and segmented data and also works with the data either on disk or already read into memory. It allows you to browse through the data and to select with the mouse sections of data that contain an artifact. Those time-segments are marked. Contrary to ft_rejectvisual, the ft_databrowser function does not return the cleaned data and also does not allow you to delete bad channels (though you can switch them off from visualization). After detecting the time-segments with the artifacts, you should call ft_rejectartifact to remove them from your data (when the data is already in memory) or from your trial definition (when the data is still on disk).
Noteworthy is that the ft_databrowser function can also be used to visualise the timecourse of the ICA components and thus easily allows you to identify the components corresponding to eye blinks, heart beat and line noise. Note that a proper ICA unmixing of your data requires that the atypical artifacts (e.g., electrode movement, squid jumps) are removed prior to calling ft_componentanalysis. After you have determined what the bad components are, you can call ft_rejectcomponent to project the data back to the sensor level, excluding the bad components.
Procedure
The following steps are used for visual artifact rejection
- Read the data into MATLAB using ft_definetrial and ft_preprocessing, as explained in the preprocessing tutorial
- Visual inspection of the trials and rejection of artifacts using ft_rejectvisual
- Alternatively you can use ft_databrowser and mark the artifacts manually by interactively paging trial by trial
Manual artifact rejection - display one trial at a time
The function ft_rejectvisual provides various ways of identifying trials contaminated with artifacts.
The configuration option cfg.method provides the possibility of browsing through the data channel by channel with cfg.method= 'channel', trial by trial with cfg.method = 'trial' or displaying all the data at once with cfg.method = 'summary'. The field cfg.latency determines the time window of interest with respect to the trigger signals. In the example below the whole trial is inspected as the latency is by default the whole trial).
The scaling of the plots is automatically adjusted according to the maximum amplitude over all channels. The scaling can be set using cfg.ylim. For EOG/EEG channels cfg.ylim=[-50e-6 50e-6] (50 micro Volt) is a useful scale and for the MEG channels cfg.ylim=[-1e-12 1e-12] (1 pT).
To browse through the data trial by trial while viewing all channels write:
cfg = [];
cfg.method = 'trial';
cfg.ylim = [-1e-12 1e-12];
dummy = ft_rejectvisual(cfg, data_all);
Click through the trials using the > button to inspect each trial.
If your dataset contains MEG and ExG channels like this dataset, the MEG and ExG channels are scaled differently when using only cfg.ylim (the ExG channels show up as big black bars on the screen). One of the reasons to record ExG (i.e., EOG, EMG or ECG) is to check these channels while identifying eye, muscle and heart artifacts. The following code can be used to scale MEG and ExG channels both properly:
cfg = [];
cfg.method = 'trial';
cfg.ylim = [-1e-12 1e-12];
cfg.megscale = 1;
cfg.eogscale = 5e-8;
dummy = ft_rejectvisual(cfg, data_all);
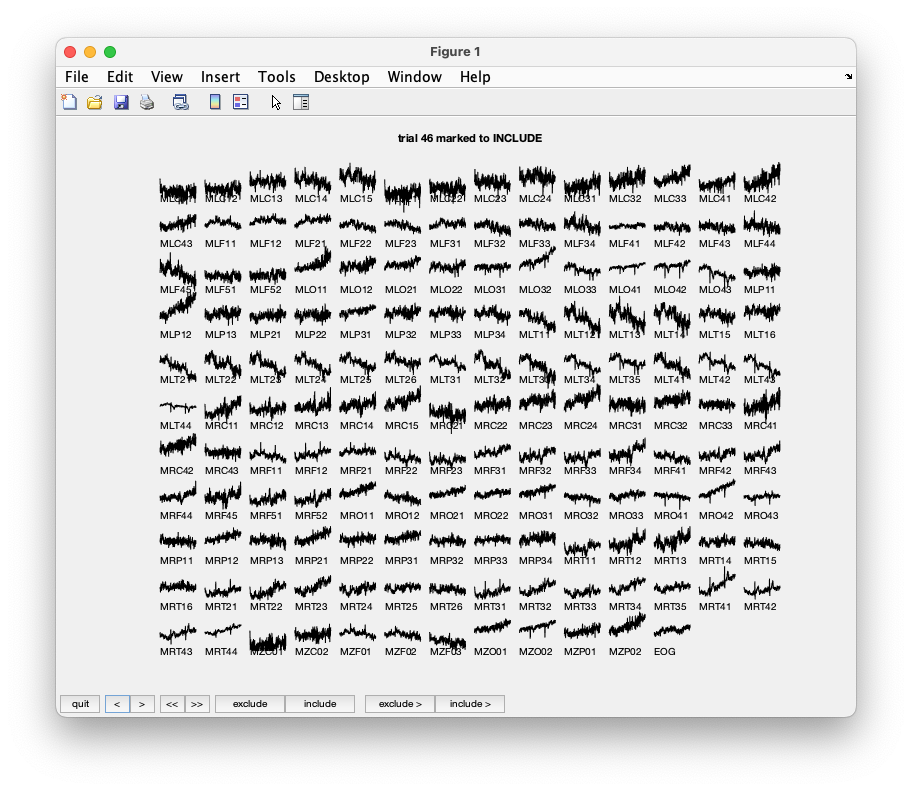
In trial 46 notice the slower drift observed over a larger group of sensors. This is most likely due to a head movement.
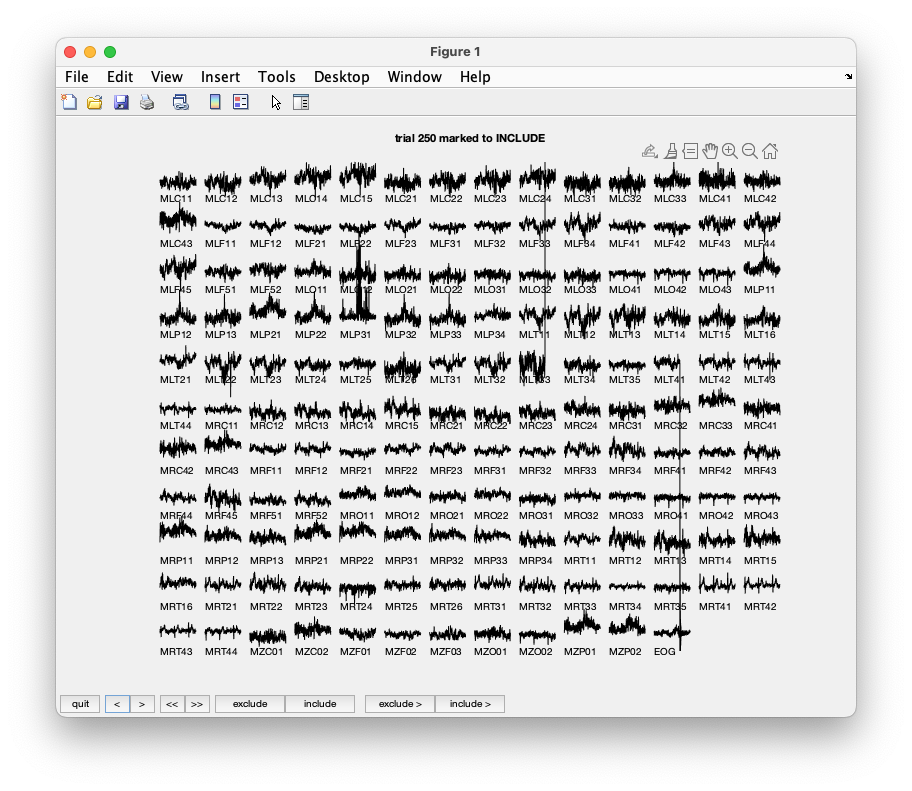
Trial 250 shows an artifact which is caused by the electronics. Notice the jump in sensor MLT41.
By browsing artifacts become evident in the trials 2, 5, 6, 8, 9, 10, 12, 39, 43, 46, 49, 52, 58, 84, 102, 107, 114, 115, 116, 119, 121, 123, 126, 127, 128, 132, 133, 137, 143, 144, 147, 149, 158, 181, 229, 230, 233, 241, 243, 245, 250, 254, 260. They should be marked as ‘bad’. After pressing the ‘quit’ button the trials marked ‘bad’ are now removed from the data structure.
If you would like to keep track of which trials you reject, keep in mind that the trial numbers change when you call ft_rejectvisual more than once. An example: There are 261 trials in your data and first you reject trial 2, 5 and 6. Then trial number 8 becomes trial number 5. Later when you also want to reject more trials you should be very careful and subtract 3 from all the old trial numbers. If you would like to know which trials you rejected, it is best to call ft_rejectvisual only once.
Manual artifact rejection - display one channel at a time
It can also be convenient to view data from one channel at a time. This can be particularly relevant for the EOG channel. To do so, write:
cfg = [];
cfg.method = 'channel';
cfg.ylim = [-1e-12 1e-12];
cfg.megscale = 1;
cfg.eogscale = 5e-8;
dummy = ft_rejectvisual(cfg, data_all);
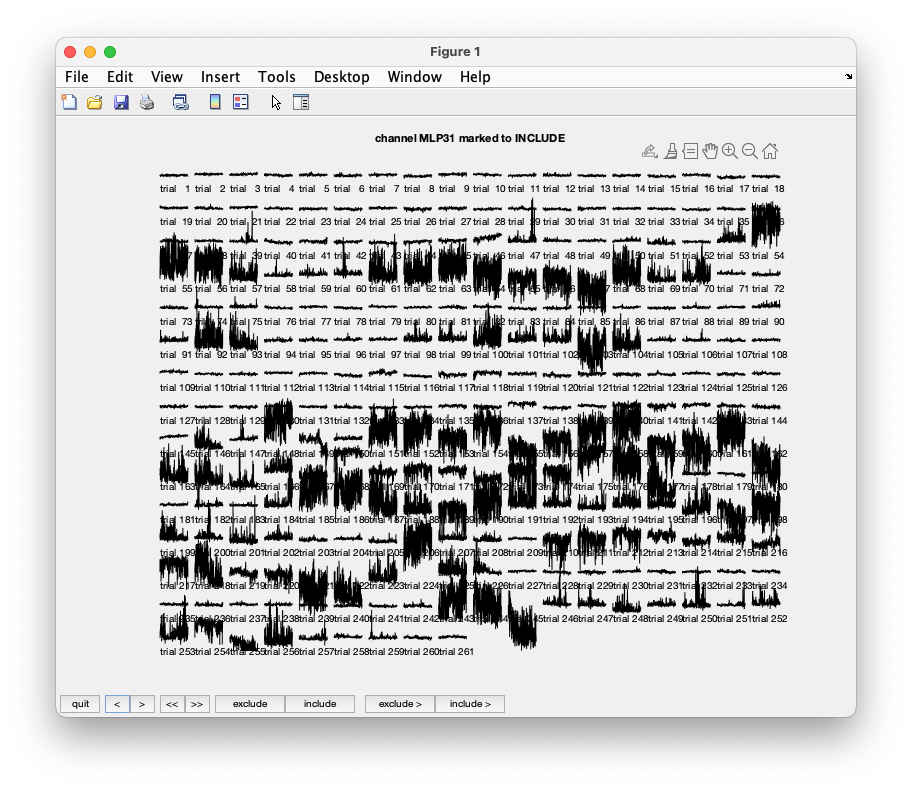
Click through the data using the > button. While clicking through all the trials you see that channels MLO12 and MLP31 contain a lot of artifacts (see the figure below ). They should be marked as ‘bad’. After pressing the ‘quit’ button the channels marked ‘bad’ are now removed from the data structure.
Manual artifact rejection - display a summary
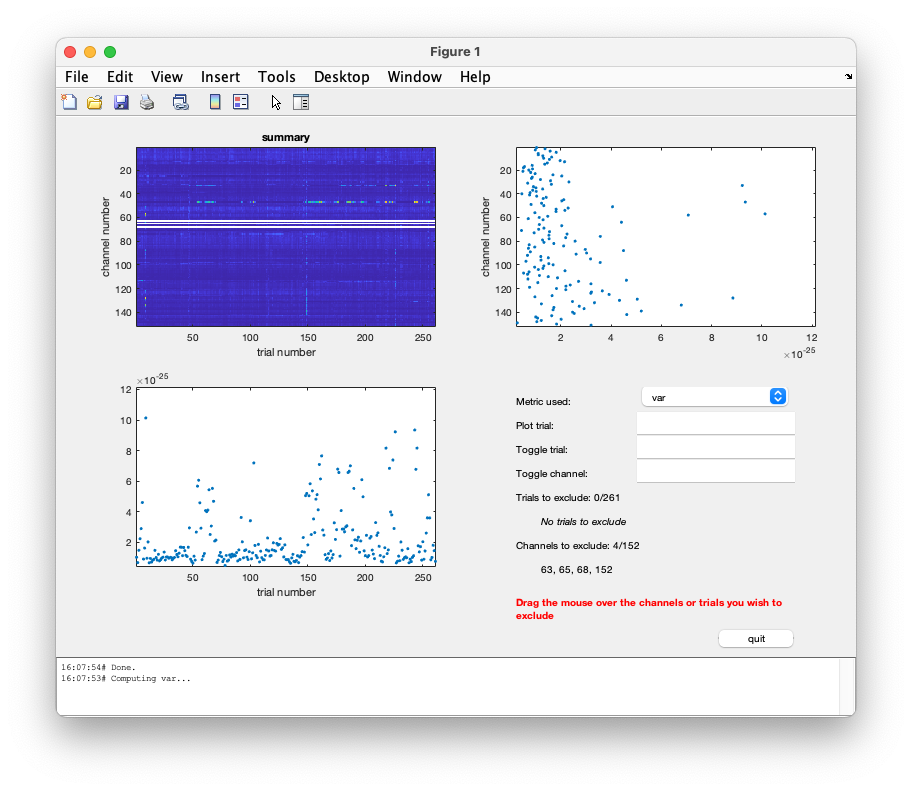
To produce an overview of the data choose the cfg.method ‘summary’:
cfg = [];
cfg.method = 'summary';
cfg.ylim = [-1e-12 1e-12];
dummy = ft_rejectvisual(cfg, data_all);
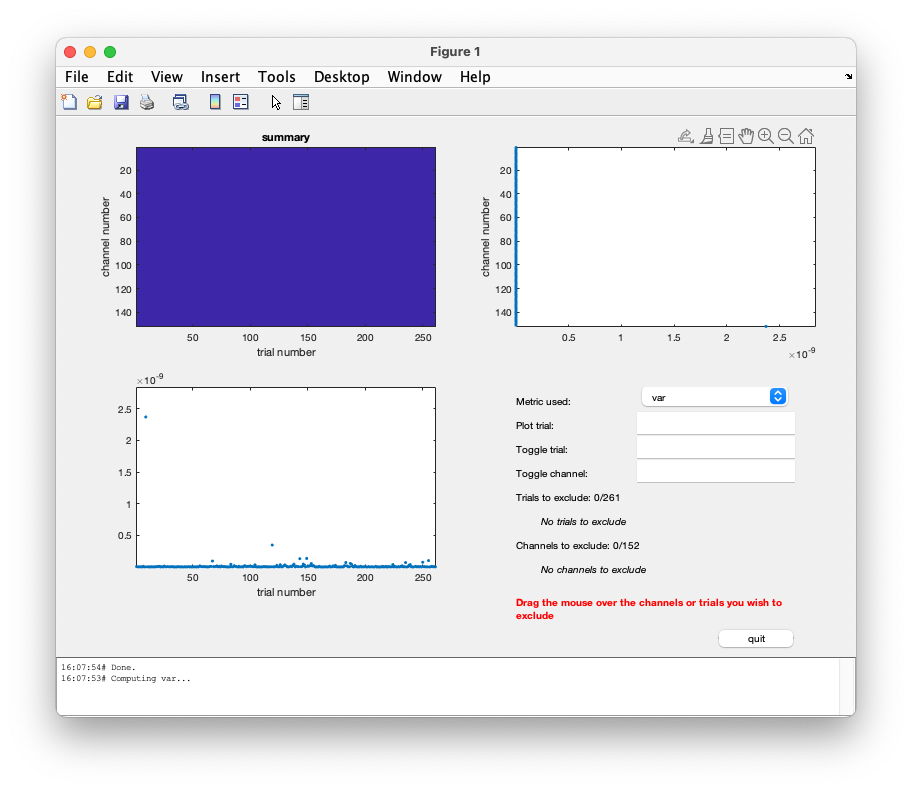
This gives you a plot with the variance for each channel and trial.
You should note that there is one channel which has a very high variance. That is the EOG channel, which contains numbers in uV which are of a very different order of magnitude than all MEG channels in T. Toggling the EOG channel will also change the figure with the maximal variance per trial (second row, left) a lot. Then you only see the variance in each trial in the MEG channels.
The command window allows for toggling trials and channels on and off. The first choice could be of toggling channels off. Enter either a channel number or its name in the edit box and enter it again to toggle it back on.
Alternatively use the mouse directly to toggle (e.g.) channels off as following: drag the mouse on the top right panel, and include the rightmost dot in the selection box. In a couple of successive steps remove the 4 channels with bigger variance and press ‘quit’.
Before pressing the ‘quit’ button, you can always toggle the channels/trials back on, by using the edit boxes ‘toggle trial’ or ‘toggle channel’.
After quitting, the trials/channels will be rejected from the data set and the command line output appears as follows:
the input is raw data with 152 channels and 261 trials
showing a summary of the data for all channels and trials
computing metric [---------------------------------------------------------]
261 trials marked as GOOD, 0 trials marked as BAD
148 channels marked as GOOD, 4 channels marked as BAD
no trials were removed
the following channels were removed: MLT31, MLT33, MLT41, EOG
the call to "ft_selectdata" took 0 seconds
the call to "ft_rejectvisual" took 243 seconds
This operation could be repeated for each of the metrics, by selecting the metric ‘var’, ‘min’, ‘max’, etc.
The summary mode in ft_rejectvisual has been primarily designed to visually screen for artefacts in channels of a consistent type, i.e., only for the axial MEG gradiometers in this example.
If you have EEG data, the EOG channels have the same physical units and very similar amplitudes and therefore can be visualised simultaneously.
If you have data from a 306-channel Neuromag system, you will have both magnetometers and planar gradiometers, which have different physical units and rather different numbers. Combining them in a single visualization is likely to result in a biassed selection, either mainly relying on the magnetometers or the gradiometers being used to find artefacts.
You can use the following options in ft_rejectvisual to apply a scaling to the channels prior to visualization: cfg.eegscale, cfg.eogscale, cfg.ecgscale, cfg.emgscale, cfg.megscale, cfg.gradscale and cfg.magscale.
You can also call ft_rejectvisual multiple times, once for every type of channels in your data. If you use cfg.keepchannel=’yes’, channels will not be removed from the data on the subsequent calls. For example:
cfg = [];
cfg.method = 'summary'
cfg.keepchannel = 'yes';
cfg.channel = 'MEGMAG';
clean1 = ft_rejectvisual(cfg, orig);
cfg.channel = 'MEGGRAD';
clean2 = ft_rejectvisual(cfg, clean1);
cfg.channel = 'EEG';
clean3 = ft_rejectvisual(cfg, clean2);
The previous example of calling ft_rejectvisual sequentially does not allow to exclude bad channels from the data structure. If youw ant to select both trials and channels, you can use the following approach:
% split the data over three different channel types
cfg = [];
cfg.channel = 'MEGMAG';
dummy1 = ft_selectdata(cfg, orig);
cfg.channel = 'MEGGRAD';
dummy2 = ft_selectdata(cfg, orig);
cfg.channel = 'EEG';
dummy3 = ft_selectdata(cfg, orig);
cfg = [];
cfg.method = 'summary';
cfg.keepchannel = 'no'; % actually this does not matter
cfg.keeptrials = 'no'; % actually this does not matter
% select channels and trials in each dataset
dummy1 = ft_rejectvisual(cfg, dummy1);
dummy2 = ft_rejectvisual(cfg, dummy2);
dummy3 = ft_rejectvisual(cfg, dummy3);
% make the final selection of channels and trials in the ORIGINAL data
cfg = [];
cfg.channel = union(dummy1.cfg.channel, dummy1.cfg.channel, dummy1.cfg.channel);
cfg.trials = intersect(dummy1.cfg.trials, dummy1.cfg.trials, dummy1.cfg.trials);
clean = ft_selectdata(cfg, orig);
Use ft_databrowser to mark the artifacts
An alternative way to remove artifacts is to page through the butterfly plots of the single trials, by using the ft_databrowser function. Call the function like
% first select only the MEG channels
cfg = [];
cfg.channel = 'MEG';
data_meg = ft_preprocessing(cfg, data_all);
% open the browser and page through the trials
cfg = [];
cfg = ft_databrowser(cfg, data_meg);
In the image below are two figures for the same trial (trial 228). As in the left figure first drag the mouse on the artifact to create dotted lines on either side of the artifact (left image). Then, as in the right figure click within the dotted lines
The resulting variable contains the field cfg.artfctdef.visual.artifact with an Nx2 matrix that represents the begin and end sample for each of the N marked sections. You can use ft_rejectartifact to reject the artifacts.
Summary and conclusion
Channels MLO12 and MLP31 are removed because of artifacts.
The following trials contain artifacts:
- 2, 5, 6, 8, 9, 10, 12, 39, 43, 46, 49, 52, 58, 84, 102, 107, 114, 115, 116, 119, 121, 123, 126, 127, 128, 132, 133, 137, 143, 144, 147, 149, 158, 181, 229, 230, 233, 241, 243, 245, 250, 254, 260
With the trials split per condition, this corresponds to the following trials:
- FIC: 15, 36, 39, 42, 43, 49, 50, 81, 82, 84
- IC: 1, 2, 3, 4, 14, 15, 16, 17, 20, 35, 39, 40, 47, 78, 79, 80, 86
- FC: 2, 3, 4, 30, 39, 40, 41, 45, 46, 47, 51, 53, 59, 77, 85
Suggested further reading
For an introduction to how you can deal with artifacts in FieldTrip in general, you should have a look at the Introduction: dealing with artifacts tutorial. As an alternative to visual artifact detection, you can do it automatically, see the automatic artifact rejection tutorial. Furthermore, you use ICA to remove artifacts from your data, this is explained in the cleaning artifacts using ICA tutorial.
More information on dealing with artifacts can also be found in some example scripts and frequently asked questions. Furthermore, this topic is often discussed on the email discussion list which can be searched like this.
Example scripts
- How to incorporate head movements in MEG analysis
- Independent component analysis (ICA) to remove ECG artifacts
- Independent component analysis (ICA) to remove EOG artifacts
- Combine MEG with Eyelink eyetracker data
- Use denoising source separation (DSS) to remove ECG artifacts
Frequently asked questions
- How can I consistently represent artifacts in my data?
- How can I interpret the different types of padding in FieldTrip?
- How can I use the databrowser?
- How does the filter padding in preprocessing work?
- I am getting strange artifacts in figures that use opacity
- What kind of filters can I apply to my data?
- Why does my TFR look strange (part I, demeaning)?
- Why does my TFR look strange (part II, detrending)?
- Why is there a residual 50Hz line-noise component after applying a DFT filter?